
This is how
the vaccine works

#InMAPFREmoreUnitedThanEver
How does it work?
How does the COVID-19 vaccine do its job in our bodies? How do our bodies react to the vaccine in order to become immune? This video briefly explains the process.

The race toward immunization
Since the outbreak of the COVID-19 pandemic, pharmaceutical laboratories and research groups around the world have been working on an effective vaccine to slow the progression of the virus. The race to find the compound has led to a peculiar situation in which new versions keep appearing, distributed in different ways around the world. At the time of writing, the main vaccines are:
Click on the name of the vaccines to see their place of origin and the countries that are using it.
Types of vaccine
mRNA vaccines
They use genetically modified RNA or DNA to create a protein that triggers an immune response.
Protein subunit vaccines
They contain parts of proteins or protein structures that are harmless but mimic the behavior of SARS-CoV-2, thus generating immunity.
Viral vector vaccines
The immune reaction occurs when coronavirus proteins develop from a genetically altered virus that does not cause the illness.
Inactivated virus vaccines
They work through an inactivated or live-attenuated virus that is not able to infect the person but is capable of triggering an immune response.
Diseases alleviated
through vaccination
The history of vaccination is long. So much so that, although we know vaccination to have come to light in the 18th century through the work of Edward Jenner, there are indications that its roots can be traced back to 200 BC. The benefits provided by this method are impossible to measure.
With the exception of access to potable water, there is no human intervention capable of competing with immunization as a means of containing disease and reducing mortality. The scientific community is still working to hunt for new vaccines.
VACCINES PREVENT 60 DEATHS PER HOUR WORLDWIDE
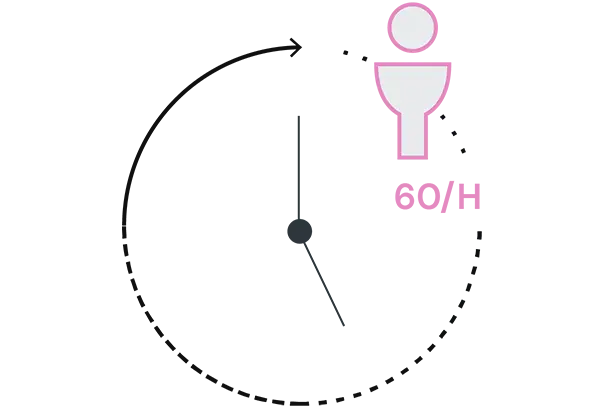
Source: OMS
They have to meet the highest quality product standards. All vaccines currently in use have undergone extensive and rigorous verification processes through constant monitoring to confirm their validity and safety.
IMMUNIZATION PREVENTS NEARLY 3 MILLION DEATHS A YEAR WORLDWIDE

Data on children under five years old. Source: Farmaindustria
What is
herd immunity?
In biostatistics, the concept of herd immunity refers to the point at which enough people in a community are protected against a disease that the spread of the disease is halted. This protection comes either from immunity due to already having had the disease or from being immunized with a vaccine. The number of people required to achieve herd immunity varies depending on the disease in question.
In the case of COVID-19, experts do not yet know what percentage of people must be vaccinated to achieve this collective immunity, but we will soon find out.
-

Healthy and non-immune people
-

Healthy and immune people
-

People infected with COVID-19
-

Successful transmission
-
Unsuccessful transmission
Initial scenario
1
When no one is immunized
2
When some people are immunized
3
When most people are immunized
Mutation, variant and strain
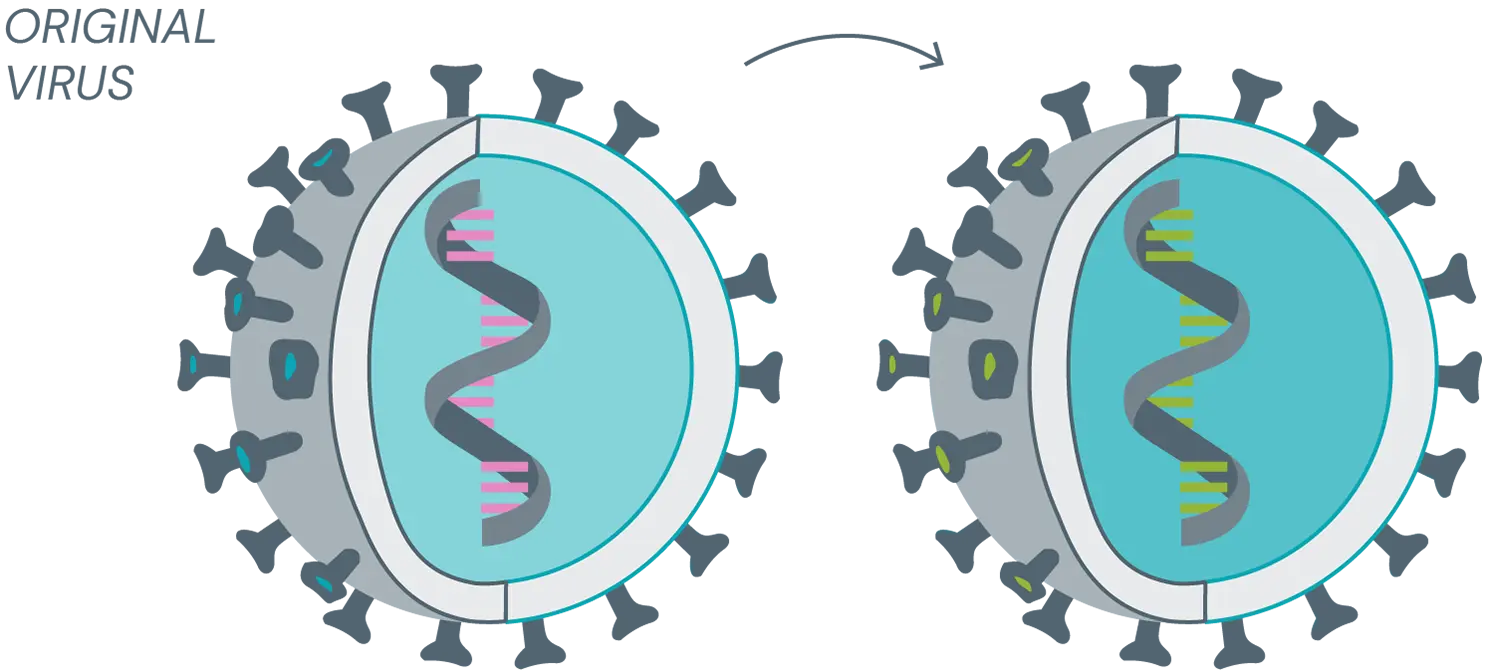
Mutation
These are subtle genetic changes that alter the characteristics of the virus, although in essence the virus remains the same. Mutations occur during virus reproduction or in the body of infected persons. The original vaccine can fight mutations.
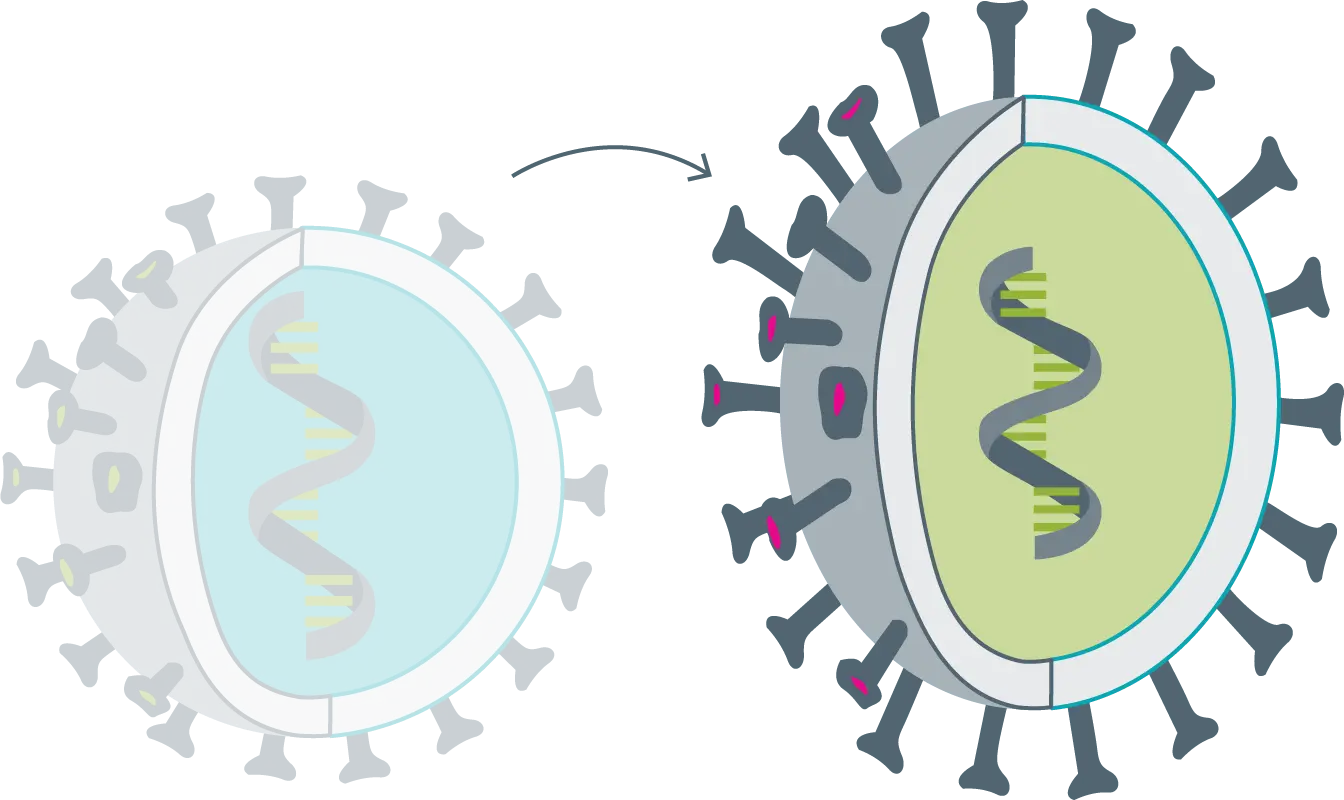
Variant
When the virus undergoes major alterations or numerous mutations. The genetic characteristics of the virus have considerably changed, but its behavior remains the same. The original vaccine still works.
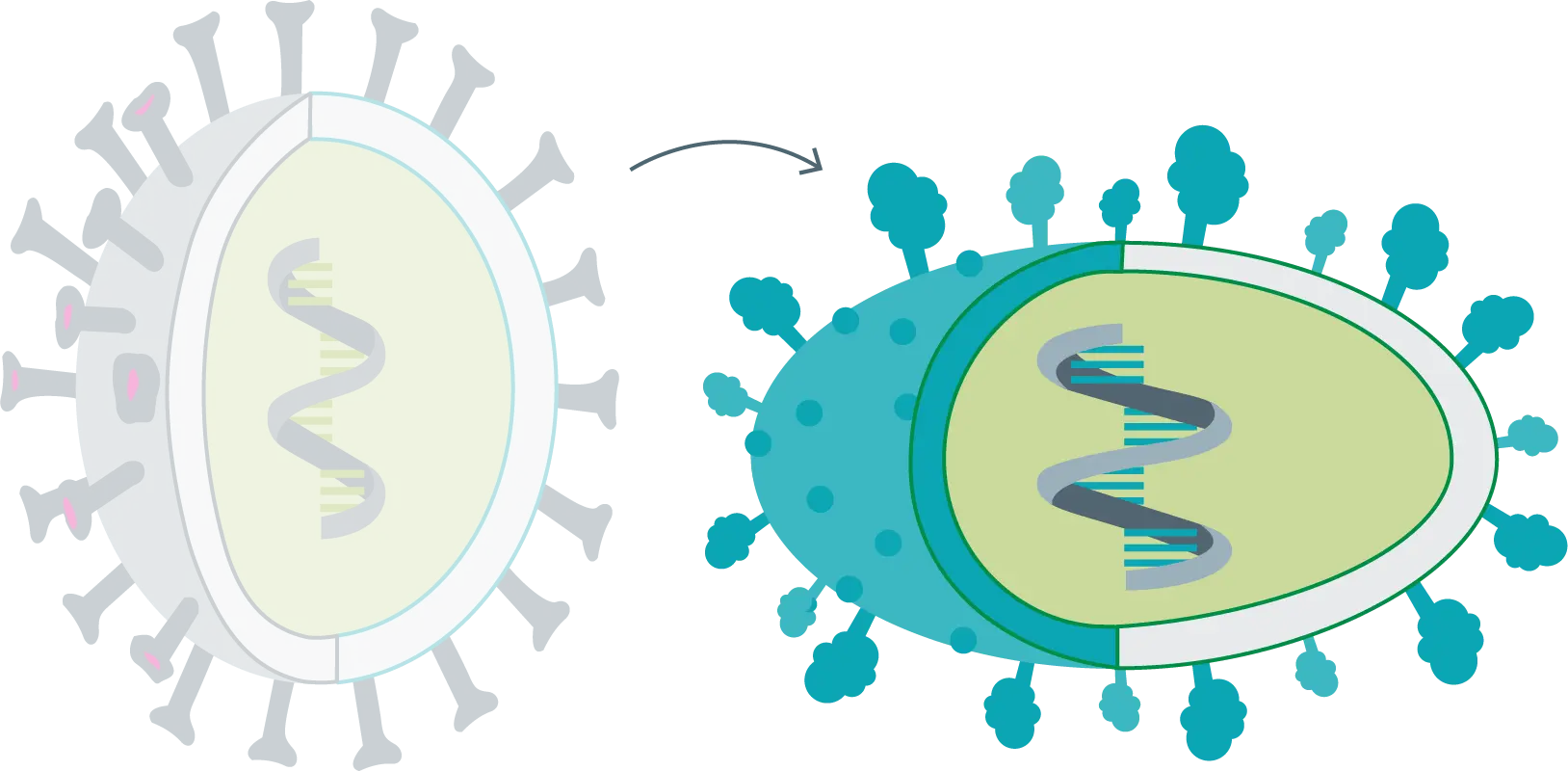
Strains
Once the virus has gone through numerous variations or mutations, there comes a time when it is difficult to match it with its origin. The characteristics of the virus have completely changed; it is almost a new virus. In these cases, the original vaccine is useful, but its effectiveness is reduced by almost half.
Vaccine myths
The rapid development of the different vaccines that are currently available has been accompanied by all kinds of information. Here, we'll clear up some misconceptions..
The vaccine modifies the genetic material of people who receive it
There is no DNA of any kind in any of the vaccines. RNA-based vaccines do not enter the cell nucleus, where our DNA is located. They are unable to modify the DNA of a cell. Vaccines do not change us in any way, except to give us immunity.
It's best to wait and see what happens with the side effects
The possibility that we could become infected with the virus, passing it on to those around us, getting sick and developing severe COVID-19 symptoms or even dying, far exceeds any possible side effects of the vaccine.
Vaccines have no effect on new variants
Although the effectiveness of approved vaccines may decrease slightly as it relates to new variants, it should be noted that some of the vaccines are up to 95 percent effective, whereas the annual flu vaccine, for example, is only 40 to 60 percent effective. That's in a good year.
It's impossible to develop safe vaccines in such a short period of time
The vaccines being used against COVID-19 have been subjected to rigorous clinical trials involving thousands of people, following initial testing on animals. The results have shown that they are safe and effective, and the data is reviewed on an ongoing basis.
You can get COVID-19 through the vaccine
The vaccines that are currently available do not use the live virus that causes COVID-19. It is necessary to keep in mind, however, that it takes a few weeks for the body to generate immunity after receiving the vaccine, so it is possible to become infected just before or just after the vaccine is administered. Or, far less likely, between doses.
Coronavirus vaccines affect fertility
This myth originated due to the fact that a small sequence of amino acids that make up one of the proteins in SARS-CoV-2 is also found in syncytin, a protein present in the placenta. However, the similarity is too short to trigger a reaction that would lead to infertility.
I am a young, healthy person — I don't need to get a vaccine
COVID-19 remains highly unpredictable, and just because someone is young and healthy doesn't mean that they won't suffer any complications. There is still no way to know whether the illness will be mild or severe, so it is far more prudent to be exposed to the vaccine than to the virus.
Vaccines contain microchips and control devices
This belief gained momentum when Microsoft co-founder, Bill Gates, talked about creating digital certificates of vaccine records. The technology that he was referring to has nothing to do with microchips. What's more, microchips are not involved in any stage of the vaccine.
We consulted the following sources when writing this special feature: The story of the vaccines, BBC, Medical News Today, The Conversation, Mayo Clinic, Cedars Sinai, Sputnik V, Cinco Días, OMS, The New York Times, Healthy Children, IS Global, Oxford Vaccine Trial.
For more information, go to vacunacovid.gob.es or the WHO website.
MAPFRE and COVID-19
MAPFRE's priority since the beginning of the COVID-19 outbreak has been protecting the health of its employees, clients and collaborators. This has been the case from that moment forward and will be into the future. That's why MAPFRE has implemented all necessary measures.
Speed up vaccination

Clients and providers
115 million euros
Donated to support SME employment and the self-employed in Spain.
60 million euros
In discounts on insurance policies, representing the value of insurance premiums that covered professional activity during the State of Emergency period.
55 million euros
In advances to our providers that were forced to stop or slow their work due to the pandemic.
Employees and collaborators
In order to ensure the safety of employees and collaborators, nearly all of them have been working remotely.
MAPFRE has not furloughed any employees, either at the company or the foundation.

Society
Because MAPFRE's commitment goes further, the company has made all SAVIA digital services available to both clients and non-clients, free of charge. This includes the availability of complete care from medical professionals through different remote channels. Chat, video consultation, email, phone.
MAPFRE also believed that COVID-19 research needed to be accelerated, so the company donated 5 million euros to the CSIC (Spanish National Research Council).
In addition, the company has extended its support to other countries, facilitating the provision of medical supplies, food and personnel to assist disadvantaged sectors.

In the USA, MAPFRE guaranteed the safety and financial burdens of its clients

In Mexico, MAPFRE acted as an agent of change

In Brazil, MAPFRE strengthened all its channels
Supporting the delivery of respirators to Manaos, the capital of the Brazilian state of Amazonas
Donating funds to Peru for the acquisition of ventilators and oxygen

Offering new protective supplies, ozone sterilization equipment and biosafety suits to Ecuador

Providing telephone support and donating 1 million euros to vulnerable populations in Mexico

Collaborating in the construction of a field hospital in Colombia
You can find more news stories in our Zoom section on mapfre.com
Fundación MAPFRE has donated more than 230,000 euros to produce a thousand pieces of equipment for a city in dire need of oxygen and with its hospitals on their knees.
COMMITMENT | 17.09.2020Extra medical and protective equipment for hospitals in HondurasFundación MAPFRE continues to ramp up the number of donations in Honduras for better equipment at two large...
HEALTH| 29.12.2020 Cities across Argentina receive donation of advanced medical equipment from Fundación MAPFRE Fundación MAPFRE has made a new donation of medical equipment in Argentina to public...




